“As a result of the feedback received by the medical and patient community and at the request of Gov. DeWine, the State of Ohio Board of Pharmacy has withdrawn proposed rule 4729:5-5-21 of the Administrative Code,” read a statement from the board. “Therefore, prohibitions on the prescribing of chloroquine and hydroxychloroquine in Ohio for the treatment of COVID-19 will not take effect at this time.”
The board will reexamine the rule with the help of the State Medical Board of Ohio, clinical experts and stakeholders to determine the next step.
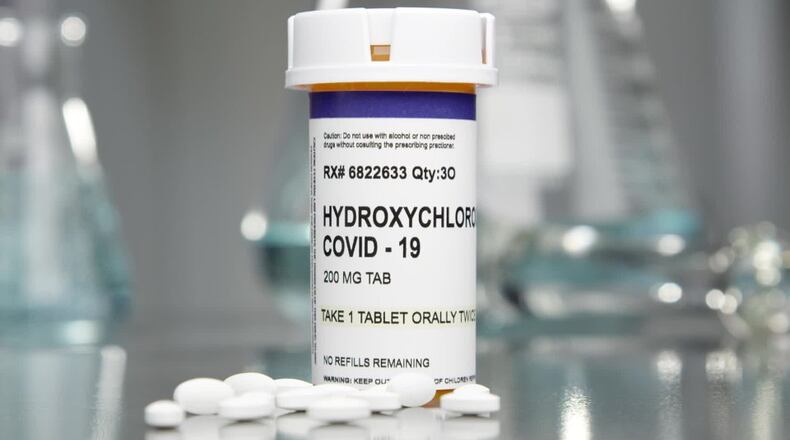
The rule, which was scheduled to go into effect today, would prohibit pharmacies from selling or dispensing hydroxychloroquine and chloroquine for coronavirus unless the use is approved by the board’s executive director. The rule would also void all previous approvals of the drug.
>> Trump floats November election delay, but it won’t happen
The governor said in a statement Thursday that he agreed with Stephen Hahn, commissioner of the Food and Drug Administration, that the decision to treat the virus with hydroxychloroquine should be between a doctor and patient.
“Therefore, I am asking the Ohio Board of Pharmacy to halt their new rule prohibiting the selling or dispensing of hydroxychloroquine or chloroquine for the treatment or prevention of COVID-19,” DeWine said. “The Board of Pharmacy and the State Medical Board of Ohio should revisit the issue, listen to the best medical science, and open the process up for comment and testimony from experts.”
The rule would not apply to prescriptions issued as part of a board-approved clinical trial to evaluate drugs to treat COVID-19. Prescriptions must include documentation that the patient is enrolled in a clinical trial.
>> Grand jury indicts Householder, 4 others in corruption case
Violations of the new rule would result in a written warning or reprimand, a fine, probation, license suspension, or permanent revolution of the license, WBNS Channel 10 reported.
Hydroxychloroquine usually is used to treat malaria, lupus and rheumatoid arthritis. Infectious disease expert Dr. Peter Hotez told KHOU Channel 11 that multiple studies have shown the drug has no proven benefit in treating or preventing the coronavirus. The FDA has revoked its emergency use on coronavirus patients, saying the risks of the drug causing serious heart problems outweigh any benefits.